2.10 |
Nucleotides and Nucleic Acids |
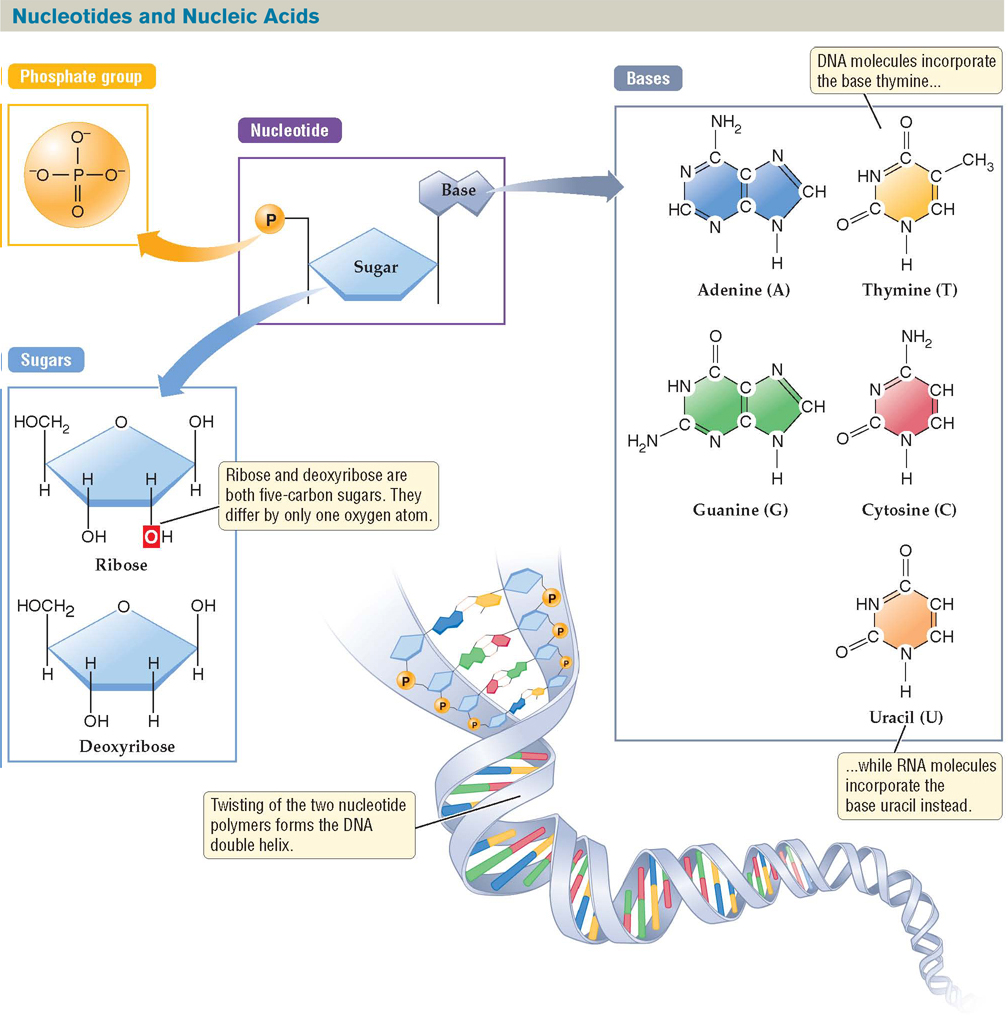
Nucleotides are important monomers in all organisms because they are the building blocks of the hereditary material. A nucleotide is a small organic molecule with three chemical components: (1) a nitrogenous base (nitrogen-containing base) that is covalently bonded to (2) a five-carbon sugar, which in turn is covalently bonded to (3) a phosphate group, a functional group consisting of a phosphate atom and four oxygen atoms (FIGURE 2.23).
FIGURE 2.23 Nucleotides Are the Building Blocks of Nucleic Acids
Each nucleotide consists of a five-carbon sugar linked to a nitrogenous base and one or more phosphate groups. The bases adenine, guanine, cytosine, and thymine, when linked to the sugar deoxyribose, form the building blocks of DNA. The bases adenine, guanine, cytosine, and uracil, when linked to the sugar ribose, form the building blocks of RNA.
Five different nucleotides—each containing one of the nitrogenous bases adenine, cytosine, guanine, thymine, or uracil (see Figure 2.23)—serve as the components for a class of polymers called nucleic acids. Nucleic acids in living cells are of two kinds: deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). DNA is distinguished from RNA both by the type of sugar in its nucleotides and by two of the nitrogenous bases that bond with that sugar. Ribose, the sugar in RNA, differs from deoxyribose, the sugar in DNA, in that it has one more oxygen atom (see Figure 2.23).
Of the five different kinds of nitrogenous bases, thymine is found only in DNA, and uracil is found only in RNA. The nucleotides in RNA and DNA are bonded through covalent linkages known as phosphodiester bonds between the sugar and phosphate groups of each successive nucleotide. The result is a chain of nucleotides, or polynucleotide. RNA consists of a single polynucleotide chain (or “strand”); DNA is composed of two polynucleotide chains (it is “double-stranded”), which are twisted in a spiral pattern to form the DNA double helix.
Nucleotides perform two essential functions in the cell: genetic information storage, and energy transfer. Every organism has nucleic acid “software” dictating how that organism will live, grow, reproduce, and respond to the external world around it. DNA can be copied in a way that preserves its sequence of nucleotides, and therefore the information coded in it.
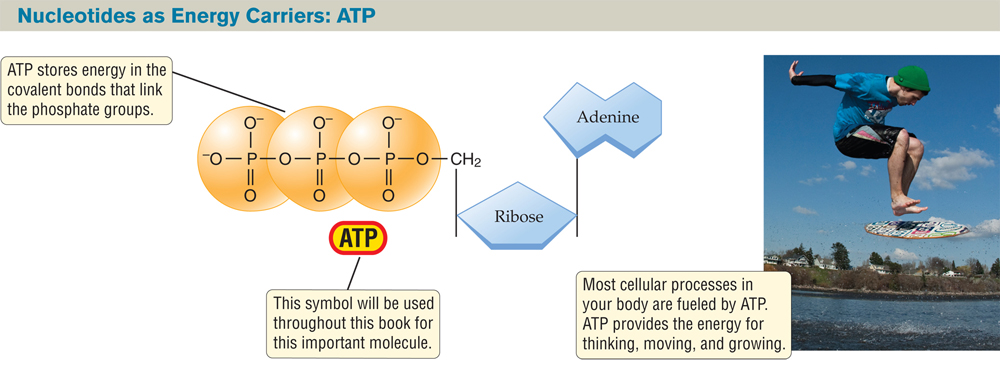
Some types of nucleotides function as energy delivery molecules, or energy carriers. The most universal of these energy carriers is the nucleotide known as adenosine triphosphate, or ATP (FIGURE 2.24). The ATP molecule is made up of adenine (A) bonded to a ribose, plus three phosphate groups. ATP is the universal energy carrier for living organisms; many cellular chemical reactions depend on energy delivered by ATP. The energy of ATP is stored in the covalent bonds that link the three phosphate groups. The breaking of the bond between two phosphate groups releases energy that is used to power other chemical reactions.
FIGURE 2.24 The Nucleotide ATP Serves as an Energy Carrier in Every Living Cell
The phosphate groups in ATP are held together by energy-rich covalent bonds. Energy is released when these bonds are broken, and the released energy powers a great variety of biological processes, including the motion of the skimboarder pictured here.
Concept Check
1. Monomers of what type are the building blocks of all nucleic acids?
Nucleotides.
2. List two differences between DNA and RNA.
DNA is a double-stranded molecule, whereas RNA is generally single-stranded. RNA has uracil instead of thymine.
APPLYING WHAT WE LEARNED
How Bad Are Trans Fats?

Back in 2003, attorney Stephen Joseph sued Kraft Foods, demanding that the food conglomerate stop selling Oreo cookies containing trans fats. At the time, most people had never heard of trans fats. But they’d heard of Oreo cookies, and Joseph’s suit was an instant sensation. Joseph didn’t care if everyone laughed at him. What mattered was that people wanted to know what trans fats were; and when they found out, they took a second, doubtful look at their Oreos.
Just days after filing the lawsuit, however, Joseph withdrew it. Media coverage of his lawsuit and of trans fats was so widespread, he said, that he could no longer argue in court that consumers didn’t know the cookies contained dangerous trans fats. Just as important, within a day of the first major news stories, Kraft reversed itself and announced it would phase out the dangerous fats. Since the “Cookie Monster” lawyer had won his point through publicity alone, he had no reason to pursue an expensive lawsuit. In fact, by the end of 2005, just 31 months later, Kraft said it had voluntarily eliminated or reduced trans fats in Oreo cookies and all of its other products.
Because saturated animal fats were thought to contribute to cardiovascular disease, most people’s grandparents and great grandparents were told that margarine and other artificial “spreads” were healthier for the heart than butter. But until recently, nearly all margarines were made from partially hydrogenated vegetable oils, which virtually always contain high levels of trans fats.
Trans fats change the ratio of two forms of cholesterol in our blood, increasing the risk of heart disease. As a result, trans fats are much worse for us than the saturated fats in butter. Experts estimate that trans fats contribute to 30,000–100,000 deaths from heart disease per year. In fact, a tiny increase in the consumption of industrial trans fats—2 percent, as measured in calories—results in a 20–30 percent increase in the risk of death from heart disease.
The U.S. Food and Drug Administration (FDA) recommends that Americans consume minimal amounts of trans fats—as little as possible. Animal fats—including those in butter, ice cream, milk, cheese, and yogurt—contain small amounts of naturally occurring trans fats, but researchers are not yet sure if these are as dangerous as the industrial trans fats in partially hydrogenated oils.
Today, nutrition labels in the United States must list how many grams of trans fats a “serving” contains. But don’t assume that “zero” means zero. If a food contains less than 0.5 gram of trans fats per serving, the manufacturer is allowed to list that as “0 g”—even if the food contains up to 0.49 gram of trans fats per serving. If a “serving” is small enough, almost any food can be listed as having “0 grams of trans fats.” As a result, it’s easy to consume enough trans fats to affect your health without knowing it. A person who ate five servings of something that contained 0.49 gram of trans fat would exceed the 2-gram daily maximum recommended by the American Heart Association. In general, it’s safe to assume that any food containing partially hydrogenated oils contains trans fats—even if the package says “Zero Trans Fats.”
BIOLOGY IN THE NEWS
For a Better, Leaner Burger, Get to Know Your Proteins
BY AMY BLASZYK ● National Public Radio (NPR), The Salt, August 17, 2012
We love our hamburgers . . . [but] all that bliss doesn’t come easy—all meat is not created equal . . .
“Meat from different species, different ages in the same species, and different cuts from the same animal will all cook differently. This is mainly because of the specific proteins in the meat as well as the fat in and around the muscle group,” [says food expert Michael Chu.]
According to Chu, there are a couple types of protein that matter in the kitchen: collagen, myosin and actin. Collagen is the fibrous—and most abundant—protein found in animals. It connects and supports body tissues, including tendons and ligaments. Myosin and actin work together to help cells move—contracting muscles . . .
So what does this all mean when it comes to grilling up a tasty burger?
When you cook meat, the collagen begins to break down into gelatin. It breaks down more slowly at low cooking temperatures, which is why you roast more fibrous cuts from the chest (brisket) or leg (shank) longer to break down the meat properly. Unfortunately, before collagen breaks down, Chu says, it does the worst thing imaginable—it tightens up. That translates into tough meat.

Chu says with burgers, the meat—and thus the collagen—is so ground up, it’s pretty much destroyed. That removes collagen from the equation, explaining why you can make a good burger out of a less tender cut, like shoulder (chuck). But now, in addition to the myosin and actin, we have something else to think about: fat. That white marbly stuff most of us spend our lives trying to lose is what makes meat in general and hamburgers in particular taste so darn good.
In cooking, fats lubricate. Using a ground meat that’s 95 percent lean will produce a dry, crumbly burger—unless you really know what you’re doing, or you compensate by adding moisture in. This is precisely the problem with turkey and venison: These two meats are naturally lean.
“Without significant fat, these burgers will naturally be lacking tenderness, flavor and succulence. Without the fat, the proteins will have very little to interfere with their natural tendencies while being heated—in particular, myosin, which, when denatured, will cross-link with each other to form a gel, like a meat glue. Too much myosin cross-linking will result in noticeable shrinkage and a rubbery texture.”
If you’re making beef burgers, Chu suggests sticking with meat that has a 20 to 25 percent fat content. With all burgers, particularly those made from leaner meats, binders like egg and breadcrumbs will do wonders.
Another suggestion from Chu if you are definitely into lean meats: browning, which binds up the proteins and will help even those leaner cuts of meat hold together. Sear your patties in a pan with a little oil before transferring them to the grill.
Cooking is a series of chemistry experiments. And creative cooking is all about clever chemistry. Take meat, poultry, and fish. Animal flesh is 70–87 percent water. The protein content of animal foods varies from 16 to 23 percent. The lipid content of muscle tissue is as low as 1 percent in lean fish to as much as 25 percent in well-marbled rib-eye steak.
The amount and distribution of proteins and lipids varies depending on the age of the animal and other physical factors, such as what it’s eating and how much exercise it’s getting. Wild-animal meat is relatively tough because game animals run around more than stockyard animals do. Lean cuts of meat tend to be tougher because they have proportionately more collagen. As noted in the news story, collagen shrinks when cooked. That’s because high temperatures denature the protein and also drive out some of the water associated with this highly hydrophilic polymer.
Marinades with low pH—those containing lemon juice, vinegar, wine, or yogurt, for example—help tenderize the meat by breaking collagen into smaller polypeptides. Protein-degrading enzymes—such as papain from papaya, bromelain from pineapples, and actinidin from kiwifruit—are also useful in meat marinades. Pounding meat, or grinding it, also breaks up the collagen, so that’s another way to tenderize it. The practice of brining—soaking in a saltwater bath for several hours—is useful for lean poultry.
Evaluating the News
1. On the basis of function, how would you classify collagen: Does it function in storage, structure, transport, or catalysis?
2. Why is pasteurized pineapple not effective in a meat marinade? (Pasteurization involves heating food to about 70°C [158°F ] to kill microbes or slow their growth.) Some people are against the pasteurization of milk. Research the topic and decide whether you agree or disagree, stating your reasons.