2.9 |
Lipids |
Lipids are hydrophobic biomolecules built from chains or rings of hydrocarbon, which, as you might guess, consists of carbon and hydrogen atoms. Fatty acids, glycerides, sterols, and waxes are examples of lipids; we will discuss these in greater detail shortly.
Most lipids are built from one or more fatty acids. A fatty acid has a long hydrocarbon chain that is strongly hydrophobic. At the other end of the hydrocarbon chain is a carboxyl group, a functional group that is polar and therefore hydrophilic. The hydrocarbon chains found in fatty acids contain many carbon atoms—16 to 22 in the lipids common in our foods. The carbon atoms in a fatty acid chain can be linked in different ways:
■ In saturated fatty acids (FIGURE 2.19a), all the carbon atoms are linked by single covalent bonds. Each carbon in the fatty acid chain is “saturated” because all of its available electrons are bonded to other atoms, which include at least two hydrogen atoms.
■ In unsaturated fatty acids (FIGURE 2.19b), one or more pairs of carbon atoms are linked by double covalent bonds. Each of these carbons is bonded to only one hydrogen atom, so the chain is not “saturated” with hydrogen.
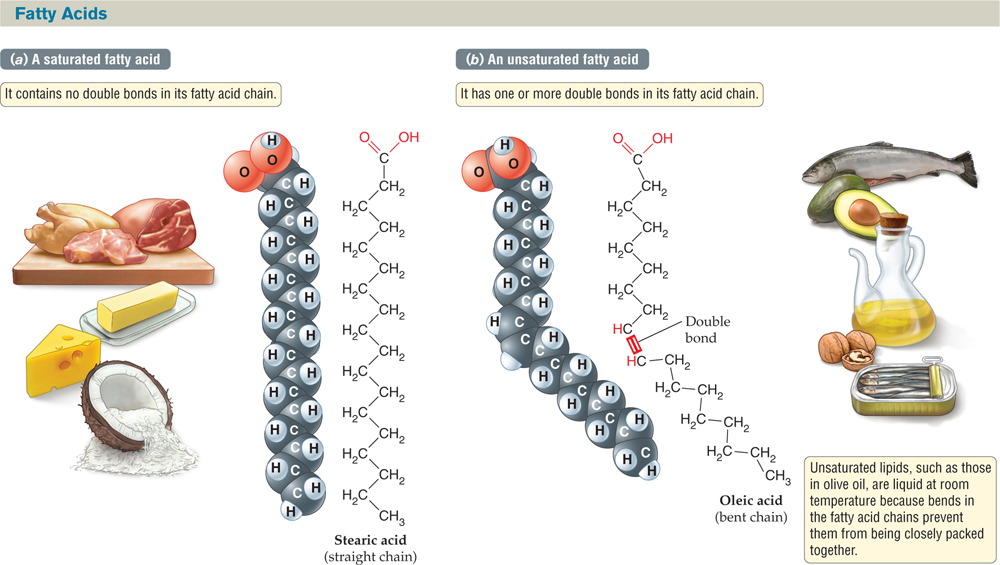
FIGURE 2.19 Saturated and Unsaturated Are the Two Main Types of Fatty Acids
The space-filling models of (a) stearic acid and (b) oleic acid show that a saturated fatty acid is a straight molecule, whereas an unsaturated fatty acid molecule has a bend in it. Saturated fatty acids can pack tightly to form a solid at room temperature, but unsaturated fats cannot.
The significance of the double bonds in unsaturated fatty acids goes beyond the difference in the number of hydrogen atoms. Saturated hydrocarbon chains tend to be straight, but the presence of double bonds in unsaturated chains can introduce kinks. The straight-chain fatty acids can pack together very tightly, forming solids or semisolids at room temperature. Unsaturated fatty acids with kinks cannot pack tightly, so these lipids tend to be liquid at room temperature.
Animals store surplus energy as triglycerides
Familiar foods such as butter and olive oil are actually complex mixtures of different types of lipids, with small amounts of other substances, such as milk protein in butter and vitamin E in most vegetable oils. The lipids in butter and olive oil include fatty acids of different types and a class of lipids called glycerides. A glyceride contains one to three fatty acids covalently bonded to a three-carbon molecule called glycerol.
Triglycerides, which consist of three fatty acids bonded to a glycerol (FIGURE 2.20), are the most common glyceride in our diet. Triglycerides built largely from saturated fatty acids tend to be solid at room temperature and are informally known as fats. Butter and lard are rich in triglycerides containing saturated fatty acids. In contrast, triglycerides rich in unsaturated fatty acids tend to be liquid at room temperature and are informally known as oils. Canola oil, olive oil, and flaxseed oil are examples, which is why all of these lipids are liquid at room temperature. Some tropical “oils,” such as those derived from coconut and palm kernels, are solid at room temperature because they also contain saturated lipids—as much as in butter and lard, or even more.
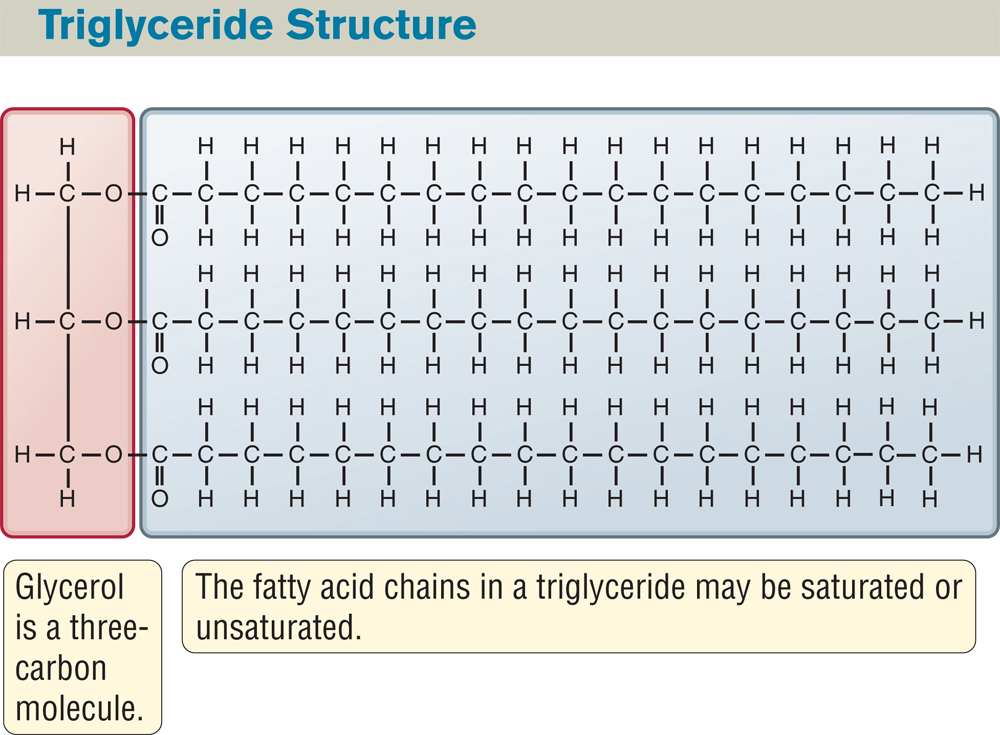
FIGURE 2.20 Triglycerides Contain Three Fatty Acids Bound to a Glycerol
Glycerides consist of a three-carbon sugar alcohol called glycerol bound to one, two, or three fatty acids. Triglycerides have three fatty acids, one linked to each of the three carbons of glycerol. The triglyceride depicted here is glyceryl tristearate, the most common storage lipid in animal cells.
A wide variety of organisms store surplus energy in the form of triglycerides, usually deposited in the cytoplasm of cells as lipid droplets. Lipids are efficient as storage reserves because they contain slightly more than twice the energy found in an equal weight of carbohydrate or protein, while occupying only one-sixth the volume. Carbohydrates and proteins take up more space inside a cell because they are hydrophilic; these macromolecules are extensively associated with water molecules, and all these extra molecules add bulk.
Phospholipids are important components of cell membranes
Phospholipids are glyceride molecules consisting of two fatty acids joined to a glycerol that bears a phosphate group. Phospholipids are major components of the plasma membrane, the outermost boundary of a cell, as well as internal cell membranes. All phospholipids have a hydrophilic “head” containing a negatively charged phosphate group, and a hydrophobic “tail” consisting of two long fatty acid chains (FIGURE 2.21a). The head group may also include other functional groups (choline in Figure 2.21a) that differ from one type of phospholipid to another.
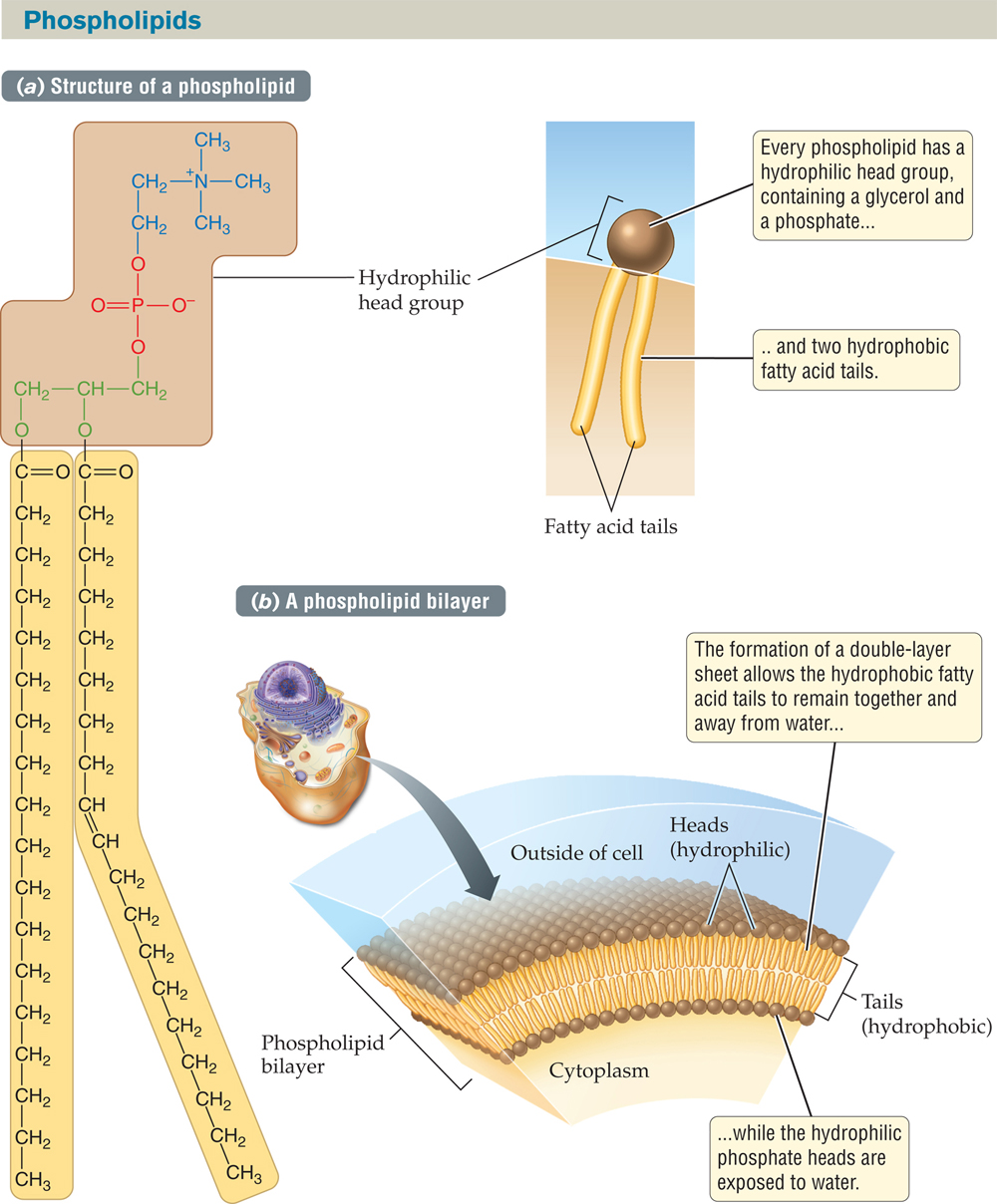
FIGURE 2.21 Membranes Contain Double Sheets of Phospholipids
(a) There are many types of phospholipids, differing in the chemistry of their head group and the length of their fatty acid tails. The one shown here is phosphatidylcholine, which has choline (blue) attached to the phosphate (red) and glycerol (green) in its head group. (b) Phospholipids spontaneously orient themselves into a double-layer sheet in which their hydrophilic head groups are oriented toward the watery environments of the cell interior and exterior, while the hydrophobic fatty acid chains convene in the middle of the “sandwich.”
Because of their dual character, phospholipids exposed to water spontaneously arrange themselves in a double-layer sheet known as a phospholipid bilayer (FIGURE 2.21b). The double layers are arranged so that the hydrophilic head groups are exposed to the watery world on either side, while the hydrophobic tails are tucked inward, away from the water. Nearly all cell membranes are organized as lipid bilayers. Cell membranes control the exchange of ions and molecules between the cells and their external environment, and also between various compartments within a cell (see Chapter 3).
Sterols play vital roles in a variety of life processes
Cholesterol, testosterone, estrogen, vitamin D—all of these are lipids with enough star quality, or medical notoriety, that they turn up on the evening news on a regular basis. Although they are widely divergent in the functions they perform, all four molecules are classified in a group of lipids known as sterols (also called steroids). All sterols have the same fundamental structure: four hydrocarbon rings fused to each other. They differ in the number, type, and position of functional groups, and in the carbon side chains linked to the four hydrocarbon rings (FIGURE 2.22).
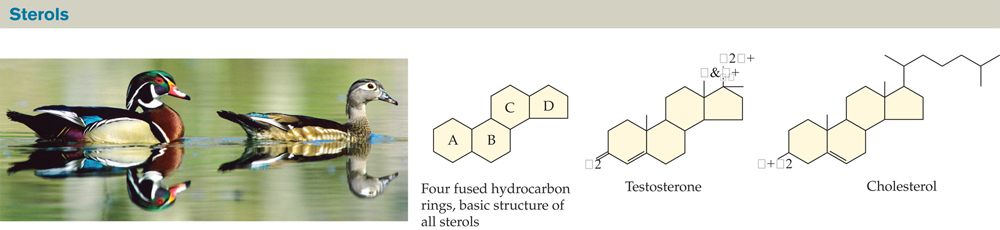
FIGURE 2.22 Sterols Are Lipids Built from Four Fused Hydrocarbon Rings
All sterols share the same basic four-ring structure but have different functional groups attached to these rings. Testosterone is a hormone that controls male sexual characteristics in many animals, including the male wood duck (photo). Cholesterol is an important constituent of the cell membranes of all birds and mammals, and of many other animals as well.
Cholesterol is the “starting” molecule for the manufacture of many other sterols, including vitamin D and bile salts; cholesterol is also a necessary component in the cell membranes of many animals. Vitamin D is important in the growth and maintenance of many tissues, especially bone and muscle. It is partially manufactured by the skin. The process is completed in the liver and kidneys. Bile salts are green, bitter-tasting lipids made by the liver and stored in the gallbladder. Bile salts aid in the digestion of fats.
Cholesterol is also the “starting” molecule in the production of steroid hormones, including sex hormones such as estrogen and testosterone. Hormones are signaling molecules that are active in very small amounts and control a great variety of processes in plants and animals. The sex hormones, such as estrogen and testosterone, promote the development and maintenance of the reproductive system in animals. Testosterone, in its several natural forms and numerous synthetic forms, is an anabolic steroid (anabolic, “putting together”). Among its many effects is the promotion of muscle growth.
The use of anabolic steroids by competitive athletes is seen as unfair advantage, and the drugs are banned by all major sports organizations. The regular use of anabolic steroids is associated with significant health risks, including higher odds of heart attack, stroke, liver damage, and liver and kidney cancer.
BIOLOGYMATTERS
Dietary Lipids: The Good, the Bad, and the Truly Ugly
Our bodies can make nearly all the lipids we need from the organic molecules we consume as food. However, a moderate intake of lipids, especially certain types of lipids, is an important part of a healthy diet. Nutritionists recommend that we consume modest amounts of unsaturated fatty acids, such as those found in olive oil and canola oil.
One class of unsaturated fatty acids, known as omega-3 fatty acids, is known for its health benefits. Flaxseed and walnuts, certain algae, and cold-water fish are good sources of omega-3 fatty acids. Ample evidence shows that EPA (eicosapentaenoic acid) and DHA (docosahexaenoic acid), the main omega-3 fatty acids in fish oil, have anti-inflammatory effects in the human body and protect against heart disease (see Chapter 1).
How saturated lipids affect human health is a complex and contentious subject. Harmful effects are seen in laboratory rats fed a diet rich in saturated lipids; the effects include increased occurrence of heart disease and some types of cancer. Studies of human populations, however, present a confusing picture, in part because human subjects are difficult to study. Some populations, such as Amish farmers and South Pacific island communities, have low rates of heart disease despite a very high intake of saturated lipids. It is possible that these populations are protected from potential negative effects because their total calorie consumption is lower or because they exercise more.
A recent analysis, which pooled data from multiple studies, suggests that the ratio of saturated to unsaturated lipids in our diet has a significant influence on disease risk. According to this report, consuming large amounts of saturated lipids, and relatively low amounts of unsaturated lipids, is correlated with greater risk of heart disease. Despite the uncertainties, most nutrition experts say that no more than 7 percent of our total calories should come from saturated fat.
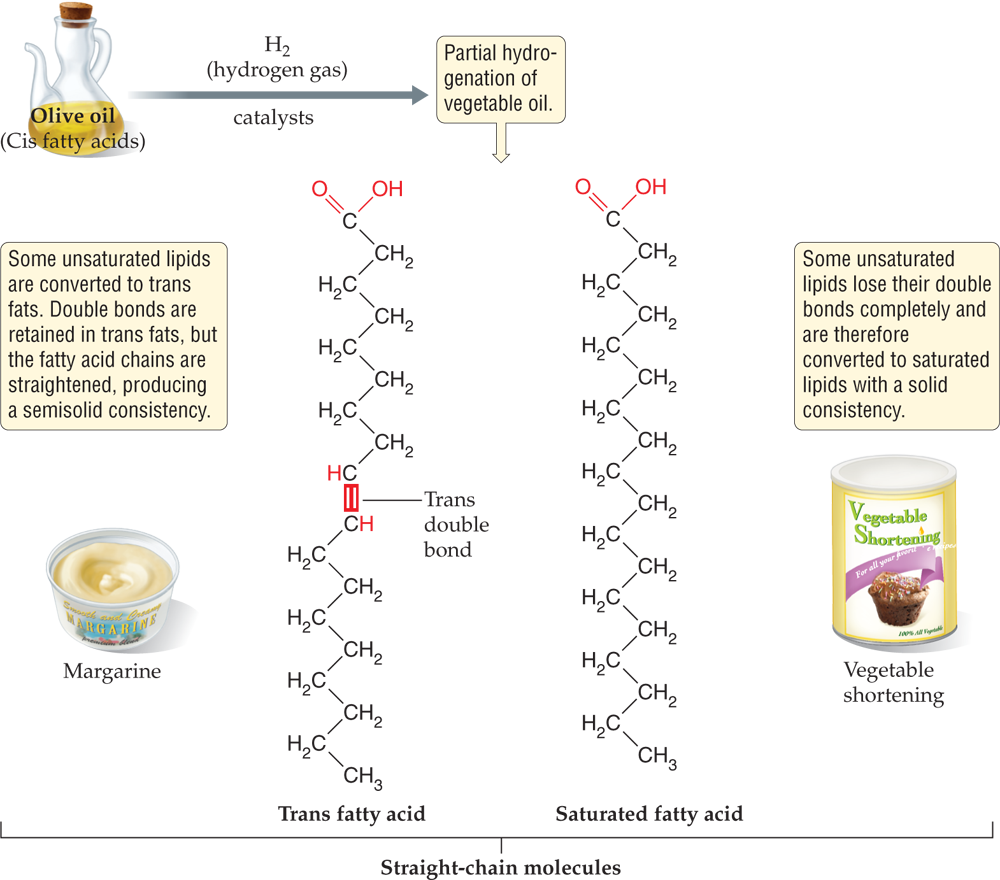
Better understood are the harmful effects of trans unsaturated lipids, known as trans fats. Trans fats contain unsaturated fatty acids whose hydrophobic tails are relatively straight compared to the bent shape of the more common (cis) type of unsaturated fatty acids. Because their straighter chains are readily compacted, trans fats are semisolid at room temperature. The overwhelming majority of trans fat in the American diet comes from partial hydrogenation of vegetable oils. The goal of this industrial process is to convert the unsaturated lipids in liquid vegetable oils to semisolid products such as margarine. As the oil is treated with hydrogen gas (H2), some of the cis fatty acid molecules are turned into saturated fatty acids through complete hydrogenation (and therefore loss) of the carbon-carbon double bonds. In some of the cis fatty acid molecules, however, the double bonds remain but swivel in a way that straightens the fatty acid tail; these straight-chain unsaturated fatty acids are trans fats.
Trans fats have been popular in the processed-food industry because they are cheaper than the alternatives and less prone to becoming rancid. Foods prepared with trans fats last well on the shelf and do not need expensive refrigeration. Cis unsaturated fatty acids, in contrast, are very susceptible to attack by oxygen gas (O2).
Concept Check
1. What is the single biggest difference between carbohydrates and lipids?
Lipids are nonpolar and therefore hydrophobic.
2. How are saturated and unsaturated fatty acids different in terms of chemical structure?
A saturated fatty acid contains only single bonds in its hydrocarbon tail; an unsaturated fatty acid contains one or more carbon-carbon double bonds.